
307 College Road East Princeton, NJ
A potential expansion site for Made Scientific's next-generation cell therapy manufacturing facility. From current Organogenesis layout to 16 ISO Class 7 GMP suites with unidirectional flow.


A potential expansion site for Made Scientific's next-generation cell therapy manufacturing facility. From current Organogenesis layout to 16 ISO Class 7 GMP suites with unidirectional flow.

A 47-second narrated walkthrough of the 307 College Road East expansion — from isometric overview through each cleanroom suite to the unidirectional material flow.
AI-Generated · Veo 3.1 video · ElevenLabs narration · 47s · 1080p
The existing 307 College Rd East layout features a cluster configuration with 45 rooms — including QC labs, cold storage, multipurpose manufacturing bays, and office space.
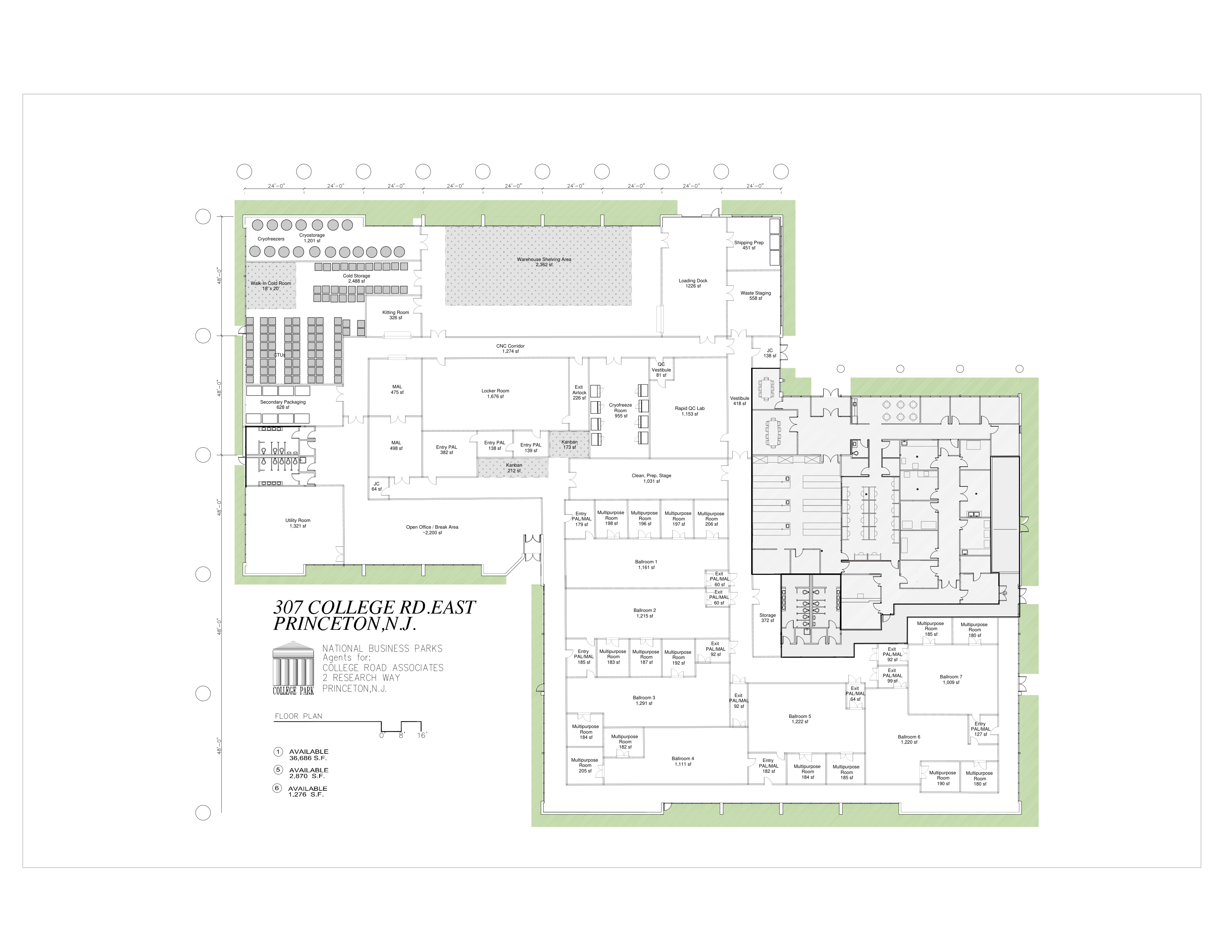
The expansion plan transforms the existing cluster layout into a purpose-built cell therapy manufacturing facility with ISO Class 7 cleanrooms, unidirectional material flow, and dedicated gowning sequences.
AI-generated 3D renders of key facility rooms, created from the original Organogenesis floor plan using Gemini 3 Pro.

Temperature-controlled storage adjacent to warehouse

QC laboratory — cell processing adjacent to Cell Room

Core QC lab — connects Egg Rm and Flask Rm

QC laboratory — downstream from Cell Room

Current manufacturing area — becomes 16 GMP suites

Quality control adjacent to manufacturing bays

Primary quality control laboratory
The expansion plan implements unidirectional flow for both materials and personnel — a GMP requirement that eliminates cross-contamination risk.


We support cell therapy programs with stage-appropriate development, GMP manufacturing, and quality systems designed to manage biological variability and enable confident clinical progression.
Early development decisions shape scalability and regulatory success. Our teams design stage-appropriate processes and analytical strategies that evolve as programs advance.
We enable programs across non-GMP, GMP clinical, and commercial manufacturing, enabling smooth transitions between development stages.
Skilled operators are the backbone of cell therapy manufacturing. We provide hands-on training programs tailored to your specific processes and quality standards.
Schedule a facility tour, request a detailed proposal, or discuss how Made Scientific's expanded manufacturing capabilities align with your cell therapy program.